Thomas E Hutson, DO, PharmD
Texas Tech University Health Science Center, School of Medicine Lubbock, Texas 79415
Editor-in-Chief, Kidney Cancer Journal
The 2026 ASCO Genitourinary Cancers Symposium, convened February 26–28 in San Francisco under the central theme of “Patient-Centered Care: From Discovery to Delivery,” delivered precisely what its title promised: a meeting that translated emerging science into clinically actionable frameworks. For those of us in the kidney cancer community, this was not merely another iteration of incremental progress. Rather, ASCO GU 2026 marks a genuine inflection point — one that reshapes how we think about the treatment continuum in renal cell carcinoma (RCC), from the post-nephrectomy adjuvant setting all the way through multiply pretreated metastatic disease.
Two late-breaking phase 3 trials — LITESPARK-011 and LITESPARK-022 — dominated the renal cell carcinoma sessions, and rightly so. Together, they represent what I believe to be the most consequential double readout for kidney cancer since the CheckMate-214 and KEYNOTE-426 era established IO-based combinations as the frontline standard. This editorial offers my perspective on what these data mean, where the unresolved questions lie, and how the broader signals from this meeting — in biomarkers, non-clear cell disease, personalization, and equity — should inform our clinical thinking and research agenda going forward.
I. Belzutifan’s Double Win: A New Therapeutic Architecture
The HIF-2α inhibitor belzutifan (Welireg) has been an intriguing molecule since its initial approval for VHL disease-associated tumors and subsequent regulatory recognition in heavily pretreated metastatic ccRCC. The intellectual foundations of this drug trace directly to Nobel Prize-winning work by Dr. William Kaelin at Dana-Farber, who elucidated the VHL-HIF pathway — a discovery that now bears fruit in daily clinical oncology. At ASCO GU 2026, we witnessed its maturation into a cornerstone agent across two distinct disease settings simultaneously, a feat that is as rare in oncology as it is significant.
LITESPARK-011: Addressing the Post-IO Void
Perhaps the most pressing unmet need in kidney cancer management has been the absence of a validated standard of care after progression on anti-PD-(L)1 therapy. VEGFR-TKIs — primarily cabozantinib — have served as de facto salvage therapy, but they were largely validated in pre-IO trial populations, raising legitimate questions about their true efficacy in a post-IO context. LITESPARK-011 was designed to answer those questions head-on.
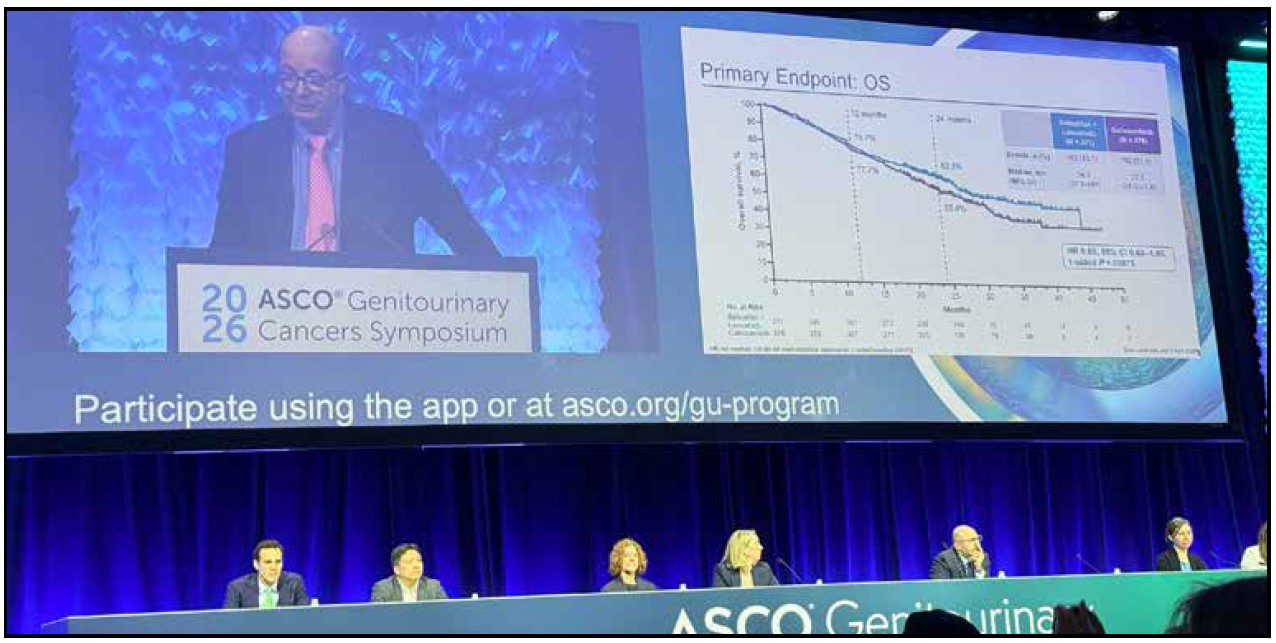
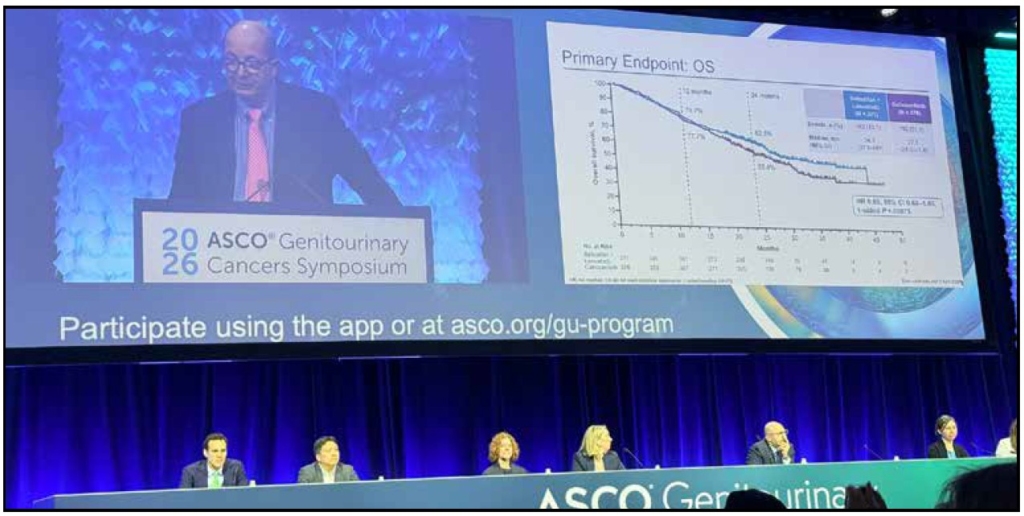
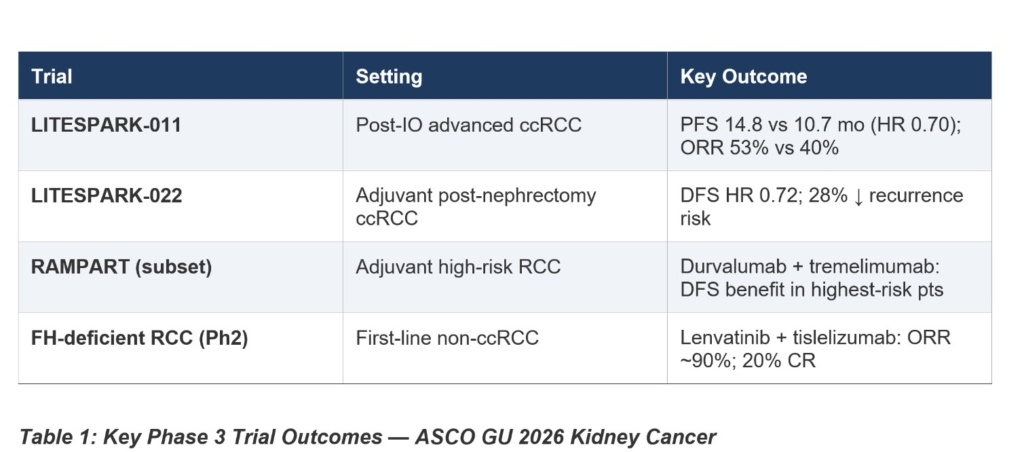
The phase 3 trial enrolled 747 patients with advanced clear cell RCC that had progressed on or after anti-PD-(L)1 therapy, randomizing them 1:1 to belzutifan plus lenvatinib versus cabozantinib monotherapy. At the second interim analysis — with a median follow-up of 29 months — the combination delivered a median PFS of 14.8 versus 10.7 months in favor of belzutifan plus lenvatinib, representing a 30% reduction in risk of progression or death (HR 0.70; p=0.00007). Equally impressive was the objective response rate: 52.6% versus 40.2%, with 20 complete responses in the experimental arm versus only 4 in the control — a signal that demands attention. Duration of response essentially doubled: 23.0 versus 12.3 months.
Overall survival numerically favored the combination — 34.9 versus 27.6 months — with a hazard ratio of 0.85, though this did not reach statistical significance at interim analysis. Dr. Robert Motzer of Memorial Sloan Kettering, the study’s lead author, appropriately framed this as an immature data point pending final analysis. Crucially, he noted that the toxicity profile was “different, not worse” than cabozantinib — anemia and hypoxia in the belzutifan arm versus diarrhea predominantly in the cabozantinib arm — and that patient-reported quality-of-life outcomes were similar between groups.
This trial is historic: it is the first phase 3 study of a HIF-2α inhibitor combined with a VEGFR-TKI, and the first such study in the post-IO RCC setting to demonstrate superiority over a contemporary VEGFR-TKI comparator. Dr. Motzer’s own comment at the pre-symposium press briefring captured the clinical gestalt precisely: for most fit patients, he would now prefer the belzutifan combination over cabozantinib in this setting. This is a statement that will reshape second-line practice rapidly and globally.
LITESPARK-022: Redefining Adjuvant Ambition
The adjuvant landscape in kidney cancer has evolved rapidly. KEYNOTE-564 established single-agent pembrolizumab as the standard of care for high-risk ccRCC following nephrectomy, demonstrating both disease-free survival and, subsequently, overall survival benefit. The natural hypothesis — tested in LITESPARK-022 — was that adding a second mechanistically complementary agent, belzutifan, could improve upon this already-validated standard.
The trial enrolled 1,841 patients — a remarkable sample size reflecting the ambition and statistical rigor of this program — randomized to pembrolizumab plus belzutifan versus pembrolizumab plus placebo for approximately one year. With a median follow-up of 28.4 months, the primary endpoint of disease-free survival was met: HR 0.72 (95% CI 0.59–0.87; p=0.0003), translating to a 28% reduction in the risk of recurrence or death. At 30 months, 75.8% of combination patients remained disease-free versus 68.6% in the pembrolizumab-alone arm — a clinically meaningful separation.
Overall survival data remain immature, with only 87 of 300 required events having occurred (HR 0.78; p=0.12). This is not a failure; it is an expected characteristic of adjuvant trials in a patient population where many are cured by surgery alone. The OS curves show early numerical separation and retain the statistical possibility of a significant benefit at final analysis. LITESPARK-022 is, notably, the first adjuvant phase 3 trial in RCC to demonstrate a significant benefit for a combination regimen versus an active immunotherapy comparator — an unprecedented benchmark.
Safety requires careful consideration. Grade 3 or higher treatment-emergent adverse events occurred in 52.1% of patients receiving the combination versus 30.2% in the pembrolizumab-plus-placebo arm. Anemia (12.1% grade ≥3), elevated ALT (6.4%), and hypoxia (4.6%) were the belzutifan-specific signals. However, grade 5 events were similar between arms (1.1% vs. 1.2%), reinforcing that the toxicity burden, while real, is manageable with appropriate monitoring and dose modification. These trade-offs will require individualized patient discussion — not every patient for whom pembrolizumab is indicated will be an appropriate candidate for the combination.
II. The Biomarker Imperative: Moving Beyond Clinical Risk Scores
One of the most intellectually rich threads running through this year’s symposium was the accelerating search for validated predictive and prognostic biomarkers in RCC. Despite the impressive efficacy gains demonstrated in LITESPARK-011 and LITESPARK-022, we continue to treat entire populations rather than selecting patients most likely to benefit — or, equally important, identifying those who will respond adequately to single-agent therapy and can be spared the toxicity of combination treatment.
Four biomarker presentations at ASCO GU 2026 stood out for their clinical potential. Kidney Injury Molecule-1 (KIM-1) — a transmembrane protein overexpressed in ccRCC whose ectodomain is detectable in peripheral blood — continues to emerge as one of the most robust circulating biomarkers in this disease. Earlier work from the IMmotion010 trial identified KIM-1 as the most reliable predictor of adjuvant therapy benefit, and new analyses presented at this symposium reinforced its dual utility as both a prognostic marker and an early indicator of treatment response to PD-1 inhibition.
Circulating tumor DNA (ctDNA) also featured prominently, with investigators from the CALYPSO trial demonstrating that baseline ctDNA levels and early ctDNA clearance carry strong prognostic value in papillary RCC patients receiving durvalumab plus savolitinib. The challenge with ctDNA in kidney cancer has always been the “low-shedding” nature of renal tumors, which limits sensitivity. Methodological refinements — including methylation-based liquid biopsy platforms — are beginning to address this limitation, and the field is watching closely.
Emerging inflammatory markers — including the neutrophil-to-eosinophil ratio, serum IgG levels, and erythropoietin — were also presented as potentially accessible, low-cost prognostic tools, particularly relevant for resource-limited settings. While none of these currently meets the bar for routine clinical implementation, they represent a growing recognition that the future of RCC management is biomarker-stratified, not population-wide.
Most forward-looking was the continuing integration of artificial intelligence and machine learning into biomarker discovery. As discussed across multiple presentations, AI has the capacity to extract patterns from imaging (radiomics), pathology slides, and multi-omic datasets that exceed human visual interpretation. The RCC field is beginning to learn from the prostate cancer experience with tools like ArteraAI, which integrates clinical and digital pathology data into actionable prognostic nomograms. We should anticipate similar frameworks emerging in RCC within the next three to five years — and we should be designing the prospective datasets now to train and validate them.
III. Non-Clear Cell RCC: Progress at the Margins, Urgency at the Center
Non-clear cell RCC — comprising papillary, chromophobe, collecting duct, fumarate hydratase-deficient, and many other rare histologies — accounts for approximately 20–25% of kidney cancer diagnoses and has historically been underrepresented in clinical trials. The consequence is stark: patients with non-ccRCC subtypes have shorter survival, fewer validated treatment options, and receive care that is, in many cases, extrapolated from clear cell data rather than grounded in subtype-specific evidence.
ASCO GU 2026 offered several encouraging signals. Most striking was updated data from a phase 2 trial in fumarate hydratase-deficient RCC evaluating lenvatinib in combination with tislelizumab (Tevimbra), which demonstrated an objective response rate approaching 90% — with 20% of patients achieving complete responses and all participants experiencing disease control. These are remarkable numbers for any solid tumor, and particularly for a subtype that has been, until recently, largely considered treatment-resistant to standard IO and TKI approaches. If validated in larger cohorts, this combination may constitute the first subtype-specific frontline standard for FH-deficient RCC.
Adjuvant pembrolizumab for non-clear cell histologies was addressed in a dedicated analysis: results showed no clear benefit compared to observation alone, reinforcing that the KEYNOTE-564 adjuvant data should not be uncritically extrapolated to non-ccRCC subtypes. A larger, molecularly stratified study is being planned. This restraint is appropriate and commendable — the non-ccRCC field must resist the temptation to borrow efficacy signals from a biologically distinct disease.
The proteogenomic characterization of non-ccRCC subtypes, including novel biomarker nominations from multi-institutional molecular profiling studies, further enriches the translational agenda for this population. Differential expression of IGF2BP3 and PYCR1 as markers of genomic instability, and the identification of multiomics signatures capable of distinguishing RCC subtypes at the molecular level, promise to improve diagnostic accuracy and therapeutic selection. These tools are not yet clinic-ready, but they are advancing at a pace that warrants attention from practicing oncologists now.
IV. The Equity and Patient-Centered Agenda
The symposium’s overarching theme — Patient-Centered Care: From Discovery to Delivery — was not merely aspirational rhetoric. The International Kidney Cancer Coalition’s global patient survey, presented as Abstract 490, provided sobering real-world data: the majority of North American patients with kidney cancer report significant emotional distress as a consequence of their diagnosis, yet few seek formal psychological support and even fewer are actively directed toward it by their clinical teams. Disparities in emotional support access were documented across racial, socioeconomic, and geographic lines.
This data point matters beyond its human significance. Psychological well-being is a determinant of treatment adherence, quality of life, and, emerging evidence suggests, immune function and oncologic outcomes. As we build more effective therapeutic regimens — regimens that are also more complex to tolerate and manage — the infrastructure to support patients holistically must scale in parallel. The oncology community cannot continue to deliver Nobel Prize-winning molecular therapy while leaving patients without access to basic mental health resources.
Equally salient is the equity dimension of personalized medicine itself. As biomarker-driven treatment selection becomes standard, we must ensure that the populations underrepresented in the trials that generated those biomarker data — including Black, Hispanic, Asian, and low-income patients — do not face a secondary exclusion from precision oncology’s benefits. The ASCO GU community is increasingly aware of this risk, and it is encouraging to see patient advocates from organizations like the IKCC participating in the scientific program, not as afterthoughts, but as collaborators in research design.
V. Looking Forward: What This Means for Practice and Research
The clinical implications of ASCO GU 2026 are clear and immediate for some questions, and still evolving for others. In second-line advanced ccRCC after anti-PD-(L)1 therapy, the data from LITESPARK-011 are sufficiently compelling that belzutifan plus lenvatinib will and should enter the therapeutic calculus for fit patients, pending regulatory action. In the adjuvant setting, LITESPARK-022 supports the consideration of pembrolizumab plus belzutifan for high-risk post-nephrectomy ccRCC patients, with the caveat that toxicity discussions must be individualized and OS data remain pending.
Sequencing questions are now the frontier. If belzutifan is used in the second line, can it still be deployed in later lines after progression? If a patient receives belzutifan adjuvantly and then recurs, what second-line options remain? These are not hypothetical future questions — they are questions our patients will ask us in clinic within the next twelve months. We need prospective trial infrastructure to answer them, and we need to build it now.
For the research community, several priorities emerge. First, the biomarker validation agenda must be formalized: ctDNA, KIM-1, and inflammatory markers need prospective validation cohorts with pre-defined endpoints and standardized assay conditions. Second, the non-ccRCC trials need to be adequately powered and molecularly enriched — small signals in rare histologies require creative trial designs, including adaptive and basket approaches, to generate actionable evidence efficiently. Third, the integration of patient-reported outcomes into trial design must become non-negotiable: the FKSI-DRS data from LITESPARK-011, showing no difference in quality-of-life deterioration between arms, is exactly the kind of information that allows patients to make genuinely informed decisions.
Finally, as Editor-in-Chief of this journal, I want to acknowledge the scientific community that made this meeting possible: the thousands of patients who enrolled in these trials trusting us with their bodies and their futures; the investigators at Memorial Sloan Kettering, Dana-Farber, and dozens of international centers who conducted this work with rigor; and the patient advocates who ensured that the questions being asked were the ones that actually matter to people living with this disease. The pace of progress in kidney cancer is accelerating. Our obligation is to ensure that it accelerates equitably, transparently, and with the patient always at the center of what we do.
Disclosures:
The author declares no conflicts of interest directly relevant to the content of this editorial.
Key References:
1. Motzer RJ, et al. Belzutifan plus lenvatinib vs cabozantinib for advanced RCC after anti-PD-(L)1 therapy: Phase 3 LITESPARK-011 Study. J Clin Oncol. 2026;44(suppl 7):LBA417.
2. Choueiri TK, et al. Adjuvant pembrolizumab plus belzutifan vs pembrolizumab for ccRCC: Phase 3 LITESPARK-022 Study. J Clin Oncol. 2026;44(suppl 7):LBA418.
3. IKCC Global Patient Survey: Emotional Concerns in Kidney Cancer and Disparities in Patient Support across North America. Abstract 490. ASCO GU 2026.
4. VHL Alliance. ASCO GU 2026 Highlights: Progress in Kidney Cancer and VHL Research. March 2026.
5. IKCC. Summary of Kidney Cancer Highlights from ASCO GU 2026. March 2026.