Kidney Cancer: Therapeutic Optimization, Biomarker Development, and Emerging Strategies
Nirmish Singla, MD, MSc
Brady Urological Institute at Johns Hopkins University

Introduction
The 2026 ASCO Genitourinary Cancers Symposium (GU26), held February 26–28 in San Francisco, California, convened leading clinicians and investigators from across the globe to present and debate the most clinically significant advances in genitourinary oncology. Kidney cancer featured prominently throughout the programme, with presentations spanning pivotal phase III trial readouts, translational biomarker research, novel combination strategies, and emerging approaches in non-clear cell histologies.
Over the past decade, the treatment paradigm for advanced renal cell carcinoma (RCC) has undergone a dramatic evolution — shifting from monotherapy with vascular endothelial growth factor (VEGF)-targeted tyrosine kinase inhibitors (TKIs) to immune checkpoint inhibitor (ICI)-based combination regimens that have meaningfully improved survival outcomes. Yet this progress has introduced new and consequential challenges: how to select among multiple effective frontline regimens, how to sequence therapy optimally after progression, and how to identify biomarkers capable of guiding individualised decision-making. GU26 2026 addressed each of these questions through updated mature follow-up data, novel mechanistic approaches, and carefully designed translational studies.
Two late-breaking phase III readouts — LITESPARK-022 and LITESPARK-011 — commanded particular attention, establishing the oral HIF-2α inhibitor belzutifan as a central pillar of RCC therapeutics across both the perioperative and advanced disease settings. Collectively, the kidney cancer sessions at GU26 reinforced the field’s trajectory toward increasingly personalised, biologically informed treatment strategies, while underscoring the continued importance of clinical trial enrolment — particularly for patients with non-clear cell histologies where the evidence base remains limited.
Evolving Frontline Therapy for Advanced Clear Cell RCC
Frontline treatment of metastatic clear cell RCC has become increasingly complex, with multiple approved immunotherapy-based combinations demonstrating durable survival benefits across risk groups. GU26 presentations provided longer-term follow-up data and comparative insights across these regimens, reinforcing the principle that no single combination offers universal superiority and that individualised regimen selection — based on disease characteristics, comorbidities, and patient preferences — remains paramount.
Dual Immune Checkpoint Blockade: Nivolumab plus Ipilimumab
Long-term follow-up data for the dual immune checkpoint blockade regimen of nivolumab plus ipilimumab continued to demonstrate durable responses and sustained survival in patients with intermediate- and poor-risk disease. Updated analyses showed that a meaningful subset of patients maintains long-term treatment-free intervals after completing the induction phase — an observation with important implications for patients who prioritise time off active treatment or who have comorbidities that limit tolerance of continuous systemic therapy.
Investigators emphasised that complete responses remain relatively uncommon but appear highly durable when achieved, with some patients maintaining remission well beyond five years without further therapy. Immune-related adverse events continue to be manageable with established corticosteroid-based algorithms, and the long-term toxicity profile was not worsened with extended follow-up. These findings reinforce dual checkpoint inhibition as a compelling frontline option for patients with lower disease burden or those for whom treatment-free intervals are a priority.
ICI-TKI Combinations: Continued Refinement
Multiple GU26 presentations examined outcomes associated with ICI-TKI combinations, spanning regimens that now constitute the most widely used frontline approaches in contemporary RCC practice:
- Pembrolizumab plus lenvatinib
- Nivolumab plus cabozantinib
- Pembrolizumab plus axitinib
Updated analyses confirmed sustained progression-free survival (PFS) and overall survival (OS) benefits across risk groups for these regimens. However, investigators noted that toxicity management and dose modifications remain important practical considerations — particularly with regimens incorporating multi-targeted TKIs such as lenvatinib or cabozantinib, where diarrhoea, hypertension, and palmar-plantar erythrodysesthesia represent frequent dose-limiting toxicities requiring proactive management and individualised dose optimisation.
An emerging theme at GU26 was the importance of moving beyond a one-size-fits-all approach to frontline selection. Speakers highlighted that patient performance status, cardiovascular comorbidities, anticipated drug-drug interactions, and the desire to preserve quality of life during treatment should all factor into regimen selection alongside the efficacy data. This nuanced perspective reflects the maturation of the field from a focus on identifying the single ‘best’ regimen toward embracing a more individualised framework.
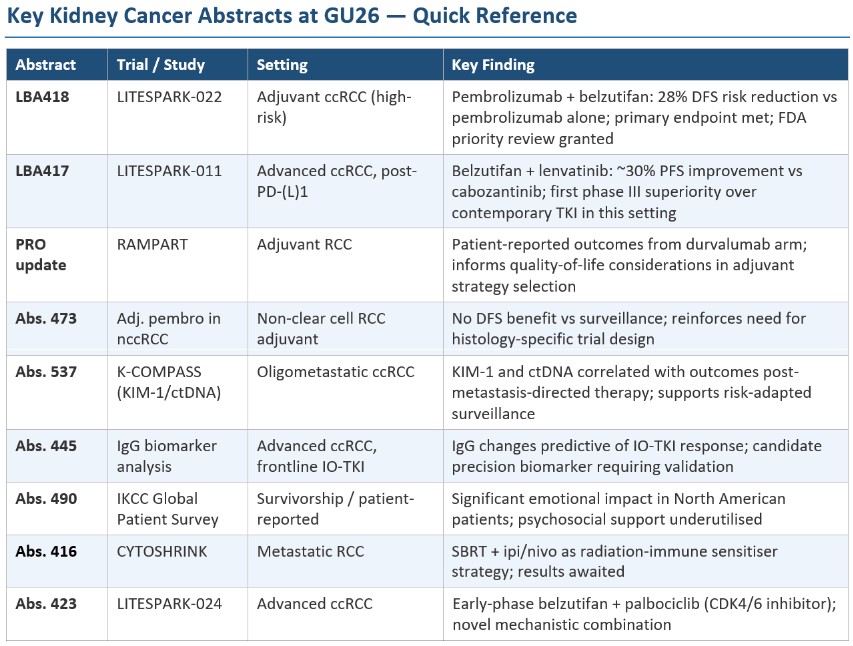
LITESPARK-022: Redefining the Adjuvant Standard in High-Risk ccRCC
Adjuvant pembrolizumab, established as standard of care based on KEYNOTE-564, has materially improved disease-free survival (DFS) in patients with high-risk ccRCC following nephrectomy. Nevertheless, a substantial proportion of treated patients still experience disease recurrence, underscoring an unmet need for more effective post-surgical strategies. LITESPARK-022 (NCT05239728) was designed to test whether adding belzutifan — an oral HIF-2α inhibitor targeting the VHL/HIF pathway that drives the majority of ccRCCs — to pembrolizumab could further extend DFS beyond what immune checkpoint blockade alone achieves.
The trial enrolled 1,841 patients with surgically resected ccRCC meeting high-risk criteria (pT2 Grade 4 or sarcomatoid; pT3 or pT4; or node-positive disease, as defined by KEYNOTE-564 eligibility). Patients were randomised 1:1 to pembrolizumab 400 mg IV every 6 weeks plus belzutifan 120 mg orally daily, or pembrolizumab plus oral placebo, for approximately one year. The primary endpoint was DFS by blinded independent central review.
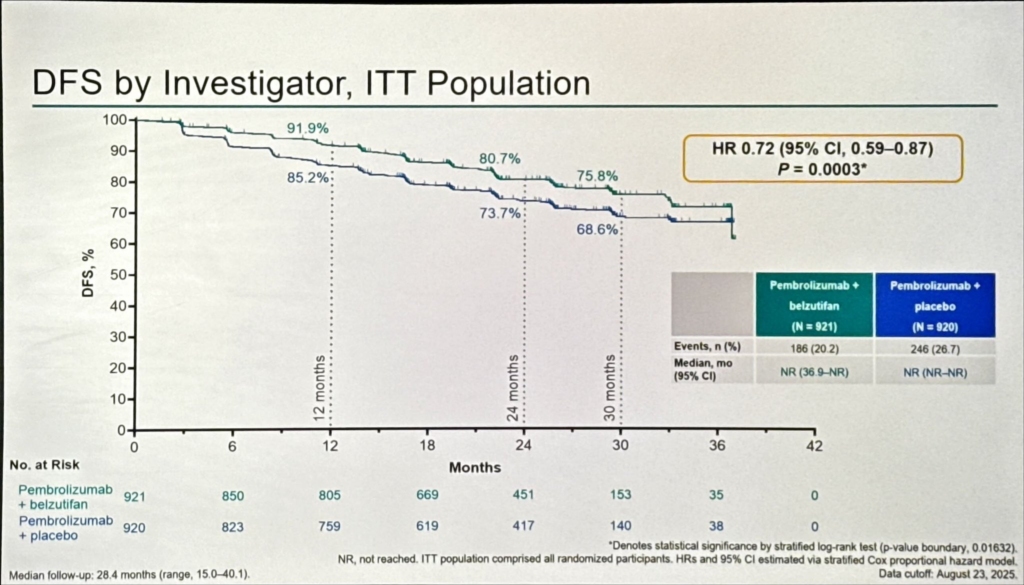
Efficacy Results
The trial met its primary endpoint with statistical significance. At a median follow-up of approximately 30 months, the combination demonstrated a 28% reduction in the risk of disease recurrence or death compared to pembrolizumab monotherapy (hazard ratio 0.72; p < 0.05). These results were presented by Dr. Toni K. Choueiri of Dana-Farber Cancer Institute, who characterised the findings as representing several simultaneous milestones: the first positive late-stage data for belzutifan in an earlier-stage disease setting; the first HIF-2α inhibitor to demonstrate benefit in combination with immune checkpoint blockade; and the first combination regimen to demonstrate superiority over pembrolizumab monotherapy in the adjuvant setting across any tumour type.
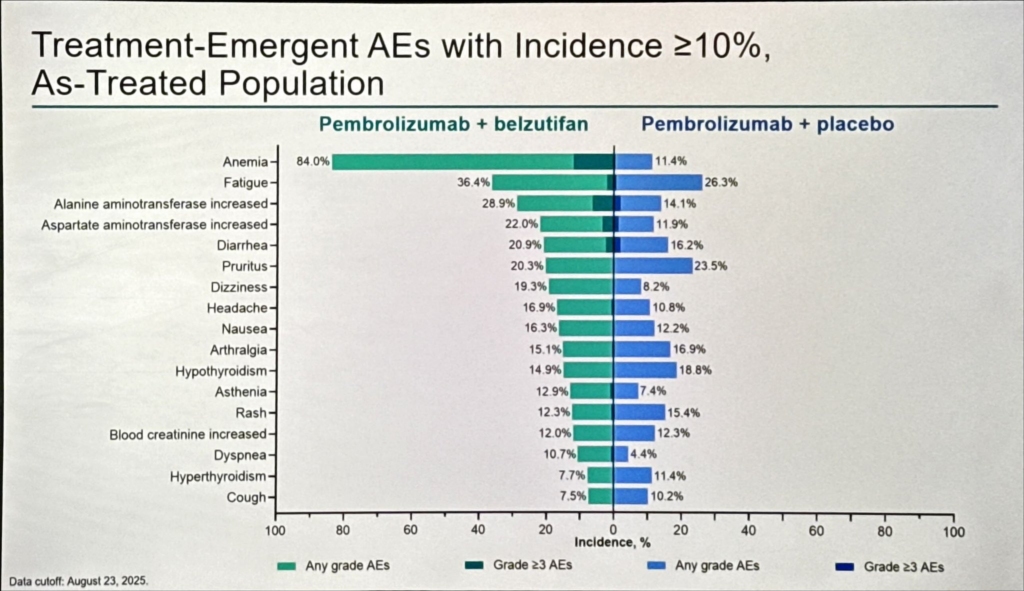
Safety and Clinical Implications
The safety profile was consistent with the known class effects of each agent. Pembrolizumab-related immune-mediated adverse events and belzutifan-related anaemia and hypoxia were the dominant toxicities; no unexpected signals emerged that would limit routine application. Monitoring of haemoglobin and oxygen saturation during belzutifan therapy remains advisable.
From a clinical practice standpoint, these data will likely shift the adjuvant standard for eligible patients. Overall survival data remain immature, and mature OS follow-up will be essential to confirm that the DFS advantage translates into a long-term survival benefit. In the interim, the magnitude of the hazard ratio reduction and the biological plausibility of the combination provide a compelling basis for regulatory approval and adoption.


LITESPARK-011: A New Post-Immunotherapy Standard in Advanced ccRCC
With the widespread adoption of ICI-based frontline regimens, clinicians increasingly face the challenge of selecting optimal therapy following progression. GU26 presentations provided important new data on this question. The failure of CONTACT-03 — which showed no benefit from adding atezolizumab to cabozantinib over cabozantinib alone — had effectively ruled out immune checkpoint rechallenge in the post-immunotherapy setting, leaving cabozantinib as the de facto standard despite the absence of a head-to-head superiority comparison in this context.
LITESPARK-011 (NCT04586231) was the first phase III trial specifically powered to test a novel doublet against cabozantinib in patients with advanced ccRCC who had progressed on anti-PD-(L)1 therapy.

Efficacy and Safety
The full data, presented by Dr. Robert J. Motzer of Memorial Sloan Kettering Cancer Center, confirmed the primary endpoint: belzutifan plus lenvatinib significantly improved PFS versus cabozantinib, reducing the risk of progression or death by approximately 30% (HR ~0.70). This represents the first phase III trial to demonstrate superiority over a contemporary TKI regimen in the post-immunotherapy ccRCC setting. A trend toward improved overall survival was observed, though OS data remained immature at this analysis.
In a notably candid exchange during the discussion, Dr. Motzer characterised the safety comparison as a ‘trade-off’ rather than an advantage for either arm. The belzutifan/lenvatinib combination produced more anaemia and hypoxia — class effects of HIF-2α inhibition — while cabozantinib produced more diarrhoea, palmar-plantar erythrodysesthesia, and hypertension.
Broader Context: Treatment Sequencing After Immunotherapy
Beyond the LITESPARK-011 data, additional GU26 presentations illuminated the broader post-immunotherapy landscape. Data confirmed that multitargeted TKIs — including cabozantinib and tivozanib — remain effective sequencing options following prior ICI exposure. Cabozantinib in particular continues to demonstrate utility attributable to its broader kinase inhibition profile, including activity against MET and AXL pathways implicated in resistance mechanisms. Emerging strategies incorporating triplet regimens or sequential HIF-2α inhibition were also discussed, though these approaches remain investigational.
Collectively, these data support the view that the second-line treatment landscape is shifting rapidly. Where cabozantinib monotherapy once stood largely unopposed, the field now has phase III evidence for a superior alternative in belzutifan plus lenvatinib, with additional options available for patients in whom the combination is contraindicated.
RAMPART: Adjuvant Immunotherapy and Patient-Reported Outcomes
The phase III RAMPART trial (NCT03288532) — investigating adjuvant durvalumab alone or combined with tremelimumab following nephrectomy — had presented its efficacy data at ESMO Congress 2025, where the durvalumab plus tremelimumab arm demonstrated a greater than 43% relative reduction in recurrence risk in the highest-risk patient subgroup. At GU26, the focus shifted to patient-reported outcomes (PROs) for the durvalumab monotherapy arm.
These data carry significant clinical weight. Adjuvant therapy decisions are increasingly informed not only by DFS curves but by the lived experience of treatment — symptom burden, functional status, and health-related quality of life during and after the treatment period. For patients already navigating the psychological aftermath of surgery for high-risk kidney cancer, the acceptability of an adjuvant regimen from the patient’s perspective is central to informed consent and adherence. The RAMPART PRO analysis provides an important evidence base that contextualises the LITESPARK-022 combination data: the community now has multiple adjuvant strategies with distinct toxicity footprints, and patient-centred outcome data will increasingly drive individualised decision-making as these regimens are adopted into practice.
Non–Clear Cell RCC: Expanding Evidence, Persistent Challenges
Non-clear cell RCC (nccRCC) subtypes — encompassing more than 20 histologically and molecularly distinct entities and collectively accounting for approximately 20–25% of all kidney cancer diagnoses — continued to receive increasing attention at GU26. Patients with nccRCC consistently experience shorter survival compared to those with ccRCC, and the absence of large prospective, histology-specific trials leaves treatment decisions underpinned by extrapolation from clear cell data and limited phase II evidence.
Adjuvant Pembrolizumab in nccRCC
Abstract 473 reported that adjuvant pembrolizumab did not demonstrate a clear disease-free survival benefit compared to active surveillance in patients with resected nccRCC. This finding is clinically important and biologically plausible: papillary RCC and other nccRCC subtypes have distinct immunological and molecular profiles, and checkpoint inhibitor benefit observed in the clear cell setting cannot be reliably extrapolated. The result reinforces the principle that nccRCC subtypes require prospective investigation in histology-specific trials — and that clinical trial participation remains strongly encouraged for patients with these diagnoses.
ICI Combinations and Emerging Data
Additional GU26 presentations included emerging data evaluating immunotherapy combinations — including checkpoint inhibitors and VEGF-targeted therapies — in papillary and chromophobe RCC, suggesting potential activity in these less common histologies. Real-world analyses in metastatic papillary RCC, where cabozantinib monotherapy and cabozantinib plus nivolumab are considered standard approaches in the absence of randomised comparisons, were also presented and will help inform the design of forthcoming cooperative group trials in this population.
Biomarkers and Precision Medicine: Progress and Remaining Gaps
Despite significant advances in systemic therapy, predictive biomarkers for treatment selection remain a critical unmet need in RCC. GU26 featured a rich programme of translational studies exploring molecular, cellular, and circulating biomarkers — reflecting the field’s recognition that the complexity of available treatment choices demands more precise patient stratification tools.
KIM-1 and ctDNA in Oligometastatic Disease
Abstract 537 presented data from the K-COMPASS programme, evaluating circulating kidney injury molecule-1 (KIM-1) protein and circulating tumour DNA (ctDNA) as response-monitoring tools in patients with oligometastatic ccRCC (up to five metastatic sites) undergoing metastasis-directed therapy — including surgery, thermal ablation, or stereotactic body radiotherapy (SBRT) — without concurrent systemic treatment. Both biomarkers correlated strongly with patient outcomes following treatment, supporting their integration with clinical parameters in the K-COMPASS risk stratification tool. Serial KIM-1 measurement may offer a non-invasive window into disease burden analogous to PSA monitoring in prostate cancer — a capability that has long been absent in kidney cancer.
Tumour Gene Expression, Immune Microenvironment, and IgG
Several translational studies explored tumour gene expression signatures and immune microenvironment characterisation as potential tools for guiding therapy selection between immune-checkpoint-dominant and VEGF-targeted strategies. Abstract 445 highlighted changes in circulating immunoglobulin G (IgG) levels as a candidate predictive biomarker for response to frontline IO-TKI combinations in advanced ccRCC, suggesting that baseline humoral immune parameters may modulate checkpoint inhibitor efficacy. Additional biomarker analyses examined the neutrophil-to-eosinophil ratio and erythropoietin levels as prognostic indicators.
Investigators uniformly cautioned that no biomarker currently demonstrates sufficient predictive accuracy for routine clinical use. The field has made meaningful progress in identifying candidate markers, but prospective validation in adequately powered, dedicated biomarker studies remains the essential next step before any of these tools can enter clinical decision-making algorithms.
Emerging Strategies: Novel Combinations and Ablative Approaches
CYTOSHRINK: SBRT as an Immune Sensitiser
The CYTOSHRINK trial, evaluating SBRT combined with ipilimumab and nivolumab in metastatic RCC, drew significant attention. The central hypothesis is that ablative doses of radiation increase tumour antigenicity through neoantigen release, priming residual metastatic disease for enhanced sensitivity to dual checkpoint inhibition. If confirmed, this multimodal approach could improve outcomes without intensifying systemic toxicity — a compelling proposition if the synergy between radiation-induced immune priming and checkpoint blockade proves durable.
Intermittent TKI Scheduling
A study examining intermittent versus continuous axitinib plus avelumab in metastatic RCC attracted interest as a quality-of-life-driven investigation into TKI scheduling. Continuous VEGFR TKI therapy imposes substantial toxicity burden, and even modest dose reductions can affect efficacy. The precedent of intermittent androgen deprivation in prostate cancer — where non-inferiority was demonstrated with meaningful quality-of-life preservation — has encouraged analogous enquiry in RCC. These data will be closely evaluated for whether treatment breaks can be safely offered without compromising PFS.
Novel Targeted Combinations: LITESPARK-024 and Beyond
Early-phase data from LITESPARK-024, evaluating belzutifan in combination with the CDK4/6 inhibitor palbociclib in ccRCC, exemplify the next generation of mechanistically-informed doublets being explored as the field moves beyond the IO-TKI paradigm. Ongoing studies are evaluating belzutifan in combinations with VEGF-targeted therapies and immune checkpoint inhibitors in earlier lines of therapy, with the goal of defining an expanded role for HIF-2α inhibition across the treatment continuum.
Patient-Centred Data and Global Perspectives
Abstract 490 presented findings from the IKCC Global Patient Survey, documenting the emotional and psychosocial impact of kidney cancer across North American patients. The data revealed that while the majority of patients experienced significant emotional distress following diagnosis, fewer than expected proactively sought psychological support or discussed these concerns with their treating oncologists — highlighting a gap between patient need and clinical practice that the oncology community must address.
The meeting also addressed the disproportionate impact of kidney cancer on older adults, with approximately 70% of new cancer diagnoses projected to occur in patients aged 65 or older by 2030. Clinical trial eligibility criteria and treatment algorithms designed around younger, fitter populations may not translate directly to this cohort, where frailty, polypharmacy, and comorbidities demand individualised assessment that goes beyond standard performance status scoring.
Clinical Implications: Key Themes from GU26
Collectively, the kidney cancer presentations at GU26 reinforced several themes shaping contemporary RCC management:
- Immunotherapy remains the backbone of frontline therapy, with multiple effective combinations available — and no single regimen offering universal superiority across all patient populations.
- Treatment selection should be individualised, balancing efficacy data, toxicity profiles, comorbidities, and patient preferences rather than defaulting to algorithmic regimen assignment.
- Novel biologic targets — most notably HIF-2α — are expanding the therapeutic landscape across both perioperative and advanced disease settings, with LITESPARK-022 and -011 providing the clearest evidence yet of this class’s broad clinical impact.
- Biomarker development remains a critical unmet need. Candidates such as KIM-1, ctDNA, IgG, and tumour gene expression signatures show promise but require prospective validation before entering routine practice.
- Non-clear cell RCC demands dedicated, histology-specific trials. Extrapolating clear cell data to nccRCC is unreliable, as the negative adjuvant pembrolizumab finding in nccRCC illustrates, and clinical trial enrolment should be prioritised for this population.
Looking Ahead
GU26 2026 will be remembered as the meeting at which belzutifan graduated from a salvage-setting agent to a central pillar of RCC therapeutics. The dual positive readouts from LITESPARK-011 and LITESPARK-022 establish HIF-2α inhibition as a cornerstone deployable across both the perioperative and advanced disease settings — a therapeutic arc that few drug classes achieve so decisively within a single symposium.
Several critical questions now demand investigation. Mature overall survival data from both LITESPARK trials are eagerly anticipated: DFS and PFS benefits are meaningful, but the field rightly awaits survival data before adjudicating on the full magnitude of these combinations. The safety trade-off in LITESPARK-011 necessitates development of patient selection frameworks and toxicity management algorithms. And the question of optimal sequencing — how to integrate belzutifan/lenvatinib as a second-line standard alongside other available options — will require both prospective data and real-world experience.
Beyond belzutifan, the meeting signalled several trajectories that will shape the next three to five years: SBRT as an immune sensitiser, intermittent TKI dosing as a quality-of-life strategy, CDK4/6 inhibitor combinations, and the emergence of validated blood-based biomarkers. For non-clear cell histologies, the community must accelerate the development of histology-specific trials rather than continuing to rely on extrapolation from the clear cell paradigm.
As the number of available treatment options continues to grow, the future of RCC management will involve increasingly personalised therapeutic strategies grounded in tumour biology, patient-specific factors, and patient-reported priorities. Ongoing clinical trials will clarify optimal sequencing, identify predictive biomarkers, and define the role of new agents across the treatment continuum. As these data mature, they will further refine the paradigm and advance the goal of durable disease control for patients with kidney cancer. The Kidney Cancer Journal will continue to provide expert analysis as this rapidly evolving landscape develops.


